TGA approval and ARTG listing are mandatory to supply medical devices in Australia.
All devices must be assessed and included on the Australian Register of Therapeutic Goods (ARTG) before they can be legally marketed.Correct classification and having a local sponsor are essential first steps.
Your device’s risk class determines documentation requirements and timelines, and only a registered Australian sponsor can submit an ARTG application.Comprehensive compliance documentation reduces delays.
Full conformity evidence, Essential Principles compliance, accurate labelling, and appropriate GMDN codes help streamline TGA processing and reduce the likelihood of audits or rework.
Licensing medical devices in Australia can feel like navigating a complex regulatory maze but it doesn’t have to be.
If you’re preparing to launch a new device, understanding the Therapeutic Goods Administration (TGA) requirements and ARTG registration process is critical. Between classification rules, sponsor obligations, and compliance documentation, many businesses feel overwhelmed before they even reach the market.
This guide breaks down how to license a medical device in Australia step by step. You’ll learn:
- Who regulates medical devices
- What approvals are required
- How to stay compliant from application to post-market obligations.
And once your device is approved, working with the right advisors, including a tax accountant can help ensure your Australian operations are structured correctly from both a regulatory and financial perspective.
Before you start the ARTG application, determine your device classification and required conformity pathway, this shapes everything from fees to documentation demands. Misclassification often leads to processing delays or additional audits.
Who is the TGA?
In Australia, the Therapeutic Goods Administration (TGA) is the national regulator for medical devices and other therapeutic goods. It operates under the Department of Health and Aged Care and plays a key role in safeguarding public health.
The TGA is responsible for evaluating, licensing, and monitoring all medical devices to ensure they are safe, effective, and high quality before reaching the Australian market.
Why does the TGA matter for licensing medical devices?
If you’re planning to supply medical devices in Australia, TGA approval is essential. Medical devices must generally be included on the Australian Register of Therapeutic Goods (ARTG) before they can be legally supplied in Australia, unless a specific exemption, exclusion, or special access pathway applies.
The TGA’s regulatory framework covers the full product lifecycle, from pre-market approvals to post-market surveillance. Their standards are internationally recognised, making TGA compliance not only a legal requirement but a mark of product credibility.
P.S.: Getting TGA approval is one part and having an ABN is another. Learn how to register your ABN and start operating legally in Australia.
How the TGA Classifies Medical Devices by Risk
When it comes to licensing medical devices in Australia, the TGA follows a strict risk-based classification system. The higher the potential risk to patients or users, the more rigorous the approval process.
Getting your classification right from the start is critical; it shapes your entire regulatory path, from documentation to fees. Misclassify it, and you could face delays or rejection.
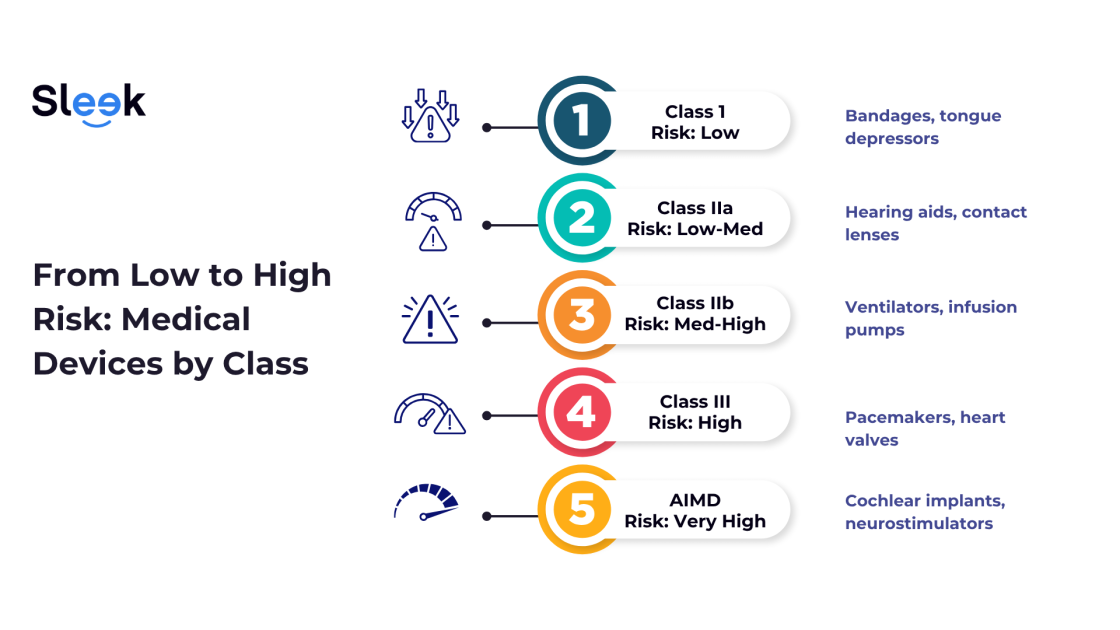
Here’s how the TGA breaks it down:
Class I: Low Risk
Basic, non-invasive items like bandages or tongue depressors.
Some may be Class Is (sterile) or Class Im (measuring), which have extra requirements.
Class IIa: Low to Medium Risk
Short-term or minimally invasive devices such as hearing aids or contact lenses.
Requires more technical documentation than Class I.
Class IIb: Medium to High Risk
Devices that support critical functions; think ventilators or infusion pumps.
Expect more in-depth assessments and evidence requirements.
Class III: High Risk
Implantable or life-sustaining devices like pacemakers or heart valves.
This class undergoes the most intensive scrutiny by the TGA.
AIMD: Active Implantable Medical Devices
Implantables that require a power source, like cochlear implants.
These sit at the highest end of the risk spectrum and face the strictest reviews.
Understanding where your device sits in this classification system is your first step toward getting it licensed and market-ready in Australia.
P.S.: The higher the risk class, the higher the stakes. Explore what business insurance you might need to stay protected.
TGA sponsor for licensing medical devices in Australia
If you’re based overseas and planning to license medical devices in Australia, you’ll need more than just a product, you’ll need a local representative. That’s where a TGA sponsor comes in.
What is a TGA sponsor?
A TGA sponsor is a registered Australian business or resident who takes legal responsibility for your device in Australia.
- They’re the ones who submit your application to the TGA, manage post-market reporting, and ensure your device stays compliant under Australian law.
- Their details appear on your ARTG certificate, so it’s a formal, accountable role, not just admin support.
From 2026, the TGA has reinforced sponsor responsibilities around importation definitions, documentation access, and ongoing compliance.
- Sponsors must ensure conformity assessment evidence and technical documentation are accessible within Australia.
- And that the device continues to meet the Essential Principles throughout its lifecycle.
Do you need a TGA sponsor to license medical devices?
Yes. If your company doesn’t have a legal presence in Australia, a TGA sponsor is mandatory. Without one, you can’t apply for TGA registration or supply your device locally.
Who can be a TGA sponsor?
The sponsor must be based in Australia, either an individual or an incorporated business. Some overseas companies use their distributor as a sponsor, but that approach can backfire.
Why an independent sponsor is often a smarter move
Your sponsor controls the ARTG listing. If things go sideways with your distributor, you don’t automatically keep your listing, they do. That means starting over, which wastes time, money, and market momentum.
Choosing an independent TGA sponsor gives you flexibility. You keep control of your regulatory pathway and can work with multiple distributors without risking your listing. It’s a cleaner, more scalable way to enter and grow in the Australian market.
P.S.: Looking to act as a sponsor or set up locally? Here’s what you need to know about getting an ACN in Australia.
Post-market obligations under TGA regulations (2026 Update)
TGA approval is not the end of your compliance obligations. Once your medical device is included in the ARTG, sponsors must meet ongoing post-market responsibilities.
These include:
Monitoring device performance and safety in the Australian market
Reporting adverse events and safety issues to the TGA within required timeframes
Maintaining access to up-to-date technical documentation and conformity evidence
Ensuring continued compliance with the Essential Principles
Complying with traceability and identification requirements where applicable (including Unique Device Identification obligations as progressively implemented)
In recent regulatory updates, the TGA has increased scrutiny on post-market vigilance and sponsor accountability. Failure to meet ongoing obligations can result in suspension, cancellation of ARTG inclusion, civil penalties, or enforceable undertakings.
Understanding these responsibilities early helps prevent costly compliance breaches and protects your long-term access to the Australian market.

What you’ll need to license a medical device in Australia
Once your device is classified and you’ve got a local TGA sponsor on board, it’s time to gather the right documentation. The TGA has a clear framework, but getting it right upfront saves you serious time later.
Step 1: Prove your device is safe via Conformity Assessment
Before your device can be listed on the ARTG, you need to show it performs safely and as intended. That’s done through a conformity assessment, a formal review of technical evidence and safety data.
For most devices (except basic Class I), this assessment must be certified either by the TGA or a recognised overseas body like a European Notified Body.
Step 2: Meet the essential principles for compliance
Every device supplied in Australia must meet a core set of rules known as the Essential Principles. These cover safety, performance, risk mitigation, labelling, and lifecycle durability.
Your evidence must show that your device:
- Performs as intended without compromising user or patient safety
- Stays safe and effective across its expected lifetime
- Minimises risk from contamination to misuse
- Is supported by clear instructions for use
To prove this, you’ll need documents like:
- Risk management reports
- Clinical evaluation summaries
- Verification and validation test results
Step 3: Use overseas evidence (if you’ve got it)
If your device already has approval from a Comparable Overseas Regulator (like CE marking or FDA clearance), you can use that as part of your TGA submission. It’s not a shortcut, but it can simplify and speed up the process.
You’ll still need to:
- Prepare a local Declaration of Conformity
- Ensure your labelling and instructions meet TGA standards
- Be prepared for a TGA audit especially for higher-risk devices
Your TGA sponsor will manage these steps and help make sure nothing’s missed.
How to get your device listed on the ARTG
The Australian Register of Therapeutic Goods (ARTG) is the official database of all medical devices legally approved for supply in Australia. Getting your device listed here is the final milestone in the licensing process.
What to expect with ARTG inclusion
The time it takes to get listed depends on:
- Your device’s classification
- The quality and completeness of your submission
- Whether your application is audited
Applications with missing documentation or unclear evidence are the most common cause of delays. Getting expert help early, especially from your sponsor, can prevent costly back-and-forth later.
ARTG listing isn’t just paperwork. It’s the green light that your device meets TGA standards and is ready for market in Australia.
Device class | Estimated timeframe
|
|---|---|
Class I (Non-sterile, non-measuring) | 1-5 days (for automatic inclusion) |
Class Is, Im, IIa, IIb (with COR evidence) | 2-3 months (if no audit is required) |
Class III & AIMD (with COR evidence) | 3-6 months (audit is likely) |
Any class requiring TGA Conformity Assessment | 6-12+ months |
What are the estimated fees for ARTG inclusion (2026)?
The TGA adjusts its fees and charges annually. The figures below are based on the TGA’s 2025-2026 schedule. You should always consult the official TGA website for the most current fee information.
1. Application fees
These fees apply when submitting an application to include a medical device in the ARTG.
Class | Application fee |
Class 3 | $1,530 |
Class 2b | $1,187 |
Class 2a | $1,187 |
Class 1 | $621 |
2. Application audit assessment fee
An application audit assessment fee may also be payable, in addition to the application fee, for certain medical devices being included in the ARTG.
Level | Application audit assessment fee |
Level 1 | $4,700 |
Level 2 | $17,288 |
P.S.: Worried about costs? Explore what government grants could help fund your device launch in Australia.
Most ARTG delays don’t happen because a device is unsafe, they happen because documentation is incomplete or misaligned. Treating compliance preparation as a priority rather than an afterthought significantly reduces approval friction.
What you’ll need to submit to the TGA?
To get your medical device listed on the ARTG, your submission needs to be complete, accurate, and audit-ready. While your TGA sponsor handles the actual submission, the manufacturer is responsible for supplying the documents.

Here’s what you’ll typically need:
- Manufacturer Evidence: Evidence of conformity assessment, such as an EU MDR certificate issued by a recognised Notified Body or certification from a Comparable Overseas Regulator (COR), where applicable. Manufacturer evidence must be submitted to and accepted by the TGA before ARTG inclusion.
- Declaration of Conformity: A legal statement confirming your device meets Australia’s Essential Principles and conformity assessment requirements.
- Labelling & IFU (Instructions for Use): Must comply with TGA standards, including sponsor details and safe-use instructions.
- Technical Documentation: A complete technical file demonstrating compliance with Australia’s Essential Principles. This includes risk management documentation (e.g. ISO 14971), clinical evidence (where required), design and manufacturing information, performance data, quality management system certification (e.g. ISO 13485), and post-market surveillance procedures. Documentation must be audit-ready.
- Device classification: The device must be correctly classified under Australian regulations (Class I, Is, Im, Ir, IIa, IIb, III, AIMD, or IVD classes). Documentation requirements and audit pathways depend on classification.
- GMDN Code: A valid and accurate Global Medical Device Nomenclature (GMDN) code must be selected. The code must precisely match the device’s intended purpose. Incorrect selection may result in delays, audit findings, or rejection of the application.
Getting this right upfront helps avoid delays and smooths your path to market approval.
What is a GMDN code and why does it matter?
The GMDN (Global Medical Device Nomenclature) code is an internationally recognised system used to classify medical devices. Each device must be assigned the correct GMDN code as part of your TGA application.
This code:
- Helps the TGA categorise and track your device
- Links your product to safety and performance data globally
- Must accurately reflect the intended use and function of your device
Choosing the right GMDN code is key. If it doesn’t match your product, your application may be delayed or rejected. Your sponsor or regulatory consultant can help confirm the best match.

Special categories: IVDs and Software-Based Medical Devices
Not all medical devices are physical products. Some are like diagnostic kits or software which fall into special categories with their own TGA rules. Misunderstanding these can put your application at risk.
IVD Registration in Australia (In Vitro Diagnostics)
IVDs are used to analyse human samples like blood or tissue. Think blood glucose monitors, HIV tests, or pregnancy kits.
The TGA uses a separate risk classification for IVDs:
- Class 1 (Low Risk) – e.g. general lab reagents
- Class 4 (High Risk) – e.g. tests for serious infectious diseases
The higher the risk of a false result, the more scrutiny you can expect. All IVDs still require a sponsor and must be listed on the ARTG before sale in Australia.
Software as a Medical Device (SaMD)
If your product is software that performs a medical function like analysing scans or calculating dosages; it’s considered Software as a Medical Device (SaMD).
The TGA assesses SaMD based on:
- What decisions it informs (e.g. life-critical vs. low-risk monitoring)
- How serious the patient’s condition is
- Whether incorrect data could harm the patient
If your software impacts urgent treatment decisions, expect a higher classification and stricter review.
Also important:
- Your software must follow the Essential Principles, including safety, performance, and risk mitigation
- You’ll need a clear privacy policy aligned with Australian law
- Cybersecurity and data protection are key, especially if patient data is involved
Getting these special cases right is essential for a smooth path to market. When in doubt, ask your sponsor or a regulatory expert before submitting.
Final steps: Importing your device and meeting ongoing TGA obligations
Getting your ARTG number is a big milestone but it’s not the finish line. Once your medical device is approved, there are still important steps to manage around importation and compliance.
Importing your device into Australia
Your TGA sponsor plays a key role here. They’re responsible for:
- Managing customs clearance
- Ensuring every shipment aligns with the ARTG listing
- Resolving any import issues before they delay your supply chain
Any mismatch in paperwork or product specs can cause costly delays. That’s why a switched-on, reliable sponsor makes all the difference.
Post-market surveillance: What happens after approval
Once your device is in the market, the work doesn’t stop. The TGA expects you and your sponsor to actively monitor your device’s safety and performance. This is part of the long-term regulatory commitment.
Ongoing obligations include:
- Reporting any adverse events to the TGA
- Handling complaints and coordinating product recalls
- Ensuring your marketing especially on social media meets TGA standards
Even after approval, the TGA can audit your product at any time. Staying on top of regulatory updates and having robust internal processes keeps your ARTG listing secure and your device available to Aussie patients.
Conclusion
Getting TGA approval might feel complex at first but with the right approach, it’s absolutely doable. It all starts with three essentials: knowing your device classification, choosing the right sponsor, and getting your compliance documents in order.
Bringing a medical device to market in Australia takes planning, precision, and patience. From risk assessments to post-market responsibilities, every step plays a role in your success.
Follow the right process, get solid advice, and you’ll be well on your way to delivering trusted health tech to the people who need it most.
Licensing a medical device isn’t just about getting a number in the ARTG, once approved, ongoing post-market responsibilities like adverse event reporting, compliance maintenance, and accurate supply documentation are mandatory under TGA regulations
TGA approval is one step. Running your Australian entity is another.
Once your regulatory pathway is underway, Sleek helps medical device businesses:
Register your Australian company correctly
Meet ASIC and corporate compliance requirements
Set up bookkeeping, payroll and financial reporting systems
Stay compliant with ATO tax obligations
Manage ongoing annual filings and governance requirements
We make sure your Australian operations are structured, compliant, and built for growth.
Start your Australian journey with the right foundation.
Answer a few quick questions and get a personalised recommendation.

450,000
businesses worldwide.
from 4,100+ reviews.
satisfaction rate from
16,000 surveyed clients.
Frequently Asked Questions
Can I switch TGA sponsors after getting ARTG approval?
You can, but it’s not automatic. The ARTG listing is tied to the sponsor, not the manufacturer. If you want to switch sponsors, you’ll need to start a new application for ARTG inclusion. That’s why using an independent sponsor (instead of your distributor) is often the smarter move long-term.
Is post-market surveillance required for Class I devices?
Yes. Even low-risk (Class I) devices are subject to ongoing post-market obligations. This includes complaint handling, adverse event reporting, and ensuring that your device continues to meet TGA compliance. It’s a common misconception that only high-risk devices need ongoing monitoring.
What happens if I choose the wrong GMDN code or device classification?
An incorrect GMDN code or classification can lead to delays, additional audits, or rejection. Your device might also be listed under the wrong category, which affects post-market compliance. Always validate your classification and code before submission, your sponsor or regulatory consultant can help with this.






